Management service organizations (MSOs) with affiliated clinical labs and urgent care clinics should take note of newly identified guardrails that will facilitate their compliance with the federal Anti-Kickback Statute (AKS). Earlier this year, the U.S. Department of Health and Human Services Office of Inspector General (OIG) issued Advisory Opinion No. 26-02, a favorable opinion concluding that an MSO could own and operate an independent clinical laboratory that accepts referrals from urgent care centers managed by the same MSO without implicating the AKS.
Based on the facts certified by the MSO, the OIG concluded that the proposed arrangement would not generate prohibited remuneration under the AKS. The opinion provides instructive guidance for MSOs, urgent care clinics, and clinical laboratories pursuing integrated care models.
The Proposed Arrangement
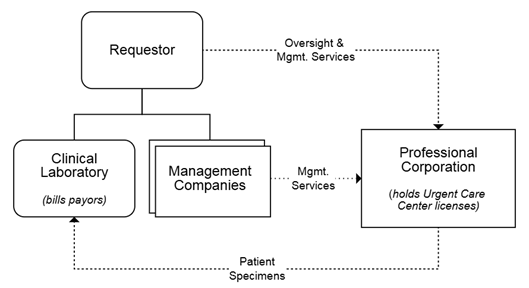
The opinion was requested by an MSO entity affiliated with four urgent care centers operated through management companies and one affiliated professional corporation. Due to state corporate practice of medicine prohibitions, the urgent care center licenses are held by the affiliated professional corporation. The MSO provides oversight and management services to the urgent care centers and holds ownership interests in the management companies associated with each urgent care center.
Under the proposed arrangement, the requestor would operate an independent clinical laboratory it would own through a separate legal entity. The laboratory would be located on premises separate from the urgent care centers. The laboratory would serve the clinical testing needs of the urgent care centers, but it would only accept specimens for testing when consistent with payor contracts and the patient’s insurance coverage. The laboratory would directly bill payors, including federal healthcare programs, for laboratory services. Neither the urgent care centers nor any providers or other facilities would be billed for laboratory services.
The requestor certified that:
- all urgent care center patients (a) would receive written notice of the affiliation between the urgent care centers and the laboratory and (b) would be offered the option to send their laboratory test to another, unaffiliated laboratory;
- no one at the urgent care centers would be required to direct testing to the laboratory, and the requestor would not track referrals from the urgent care centers to the laboratory;
- providers at the urgent care centers would be able to order from several laboratories in addition to the affiliated laboratory through the electronic health record system used by the urgent care centers with no preference given to the affiliated laboratory; and
- the laboratory would not station phlebotomists or other personnel at the urgent care centers, and all specimen collection would be performed by existing urgent care center staff.
Importantly, the requestor certified that guardrails would be in place with respect to compensation and remuneration flowing between the parties. The OIG’s analysis focused on these guardrails, which we discuss below.
Depiction of Proposed Arrangement in AO 26-02
OIG Analysis Focuses on the Remuneration Guardrails
In its analysis of the proposed arrangement, the OIG focused on three key facts certified by the requestor regarding the lack of remuneration in the arrangement:
- No part of any compensation received by providers at the urgent care centers would be tied to the volume or value of services ordered from the laboratory.
- No remuneration would flow from the laboratory, directly or indirectly, to the urgent care centers or any providers at the urgent care centers.
- The requestor would not, directly or indirectly, pay remuneration to the urgent care centers or any providers at the urgent care centers from any revenue derived from laboratory services.
Taken together, the OIG viewed these facts as a certification by the requestor that neither the requestor nor the laboratory would offer or pay any remuneration to any individual or entity in connection with the referral of specimens to the laboratory. As such, the OIG reasoned that the AKS would not be implicated because no remuneration would be paid to any individual or entity to induce referrals to the laboratory. This absence of such remuneration led the OIG to conclude that the arrangement would not violate the AKS and, therefore, would not trigger administrative sanctions by the agency.
Cautionary Notes
Although the OIG reached a favorable conclusion, it warned that similar arrangements would implicate the AKS if remuneration were paid to referral sources to induce or reward referrals to a particular laboratory for testing reimbursable by a federal healthcare program. In particular, the OIG cautioned against abusive arrangements involving management companies that own or are affiliated with clinical laboratories. The OIG identified certain arrangements it believed could be associated with “funneling” kickbacks to providers in exchange for referrals, including sham investment opportunities, sham consulting arrangements, or the provision of free personnel or equipment.
Practical Takeaways for Urgent Care and MSO Operators
This advisory opinion offers helpful guidance on how urgent care platforms can expand and integrate ancillary services without running afoul of the AKS. Based on the facts certified by the requestor, similarly situated entities should consider the following characteristics when structuring relationships with affiliated clinical laboratories:
- No remuneration of any kind. Nothing of value should be paid — directly or indirectly — to providers or facilities to induce or reward referrals of laboratory specimens. For instance, free personnel, phlebotomists, or equipment should not be provided by the laboratory, and specimen collection should performed by the provider or facility’s existing clinic staff as part of routine patient care.
- No referral steering. Providers should not be required to direct testing to an affiliated laboratory. Electronic health record systems should allow orders to multiple laboratories without preference.
- No tracking or rewarding of referrals. Managers should not track referrals from the affiliated providers to the laboratory, and provider compensation should not be tied to the volume or value of tests ordered.
- Preserve patient choice. Patients should be made aware of the affiliation between the provider and/or facility and the laboratory and should be free to choose an unaffiliated laboratory.
Akerman’s Healthcare Practice Group is available to answer questions you may have about setting up an affiliated clinical laboratory.